Vipan Kumar1, Mike Hunter2, Mike Stanyard3
1School of Integrative Plant Sciences -Soil and Crop Sciences Section, Cornell University, Ithaca, NY 14853, 2Field Crops IPM Coordinator, New York State Integrated Pest Management Program (NYSIPM), Redwood, NY, 3Cornell Cooperative Extension Northwest New York Dairy, Livestock, and Field Crops Program
As the spring weather is warming up in the New York (NY), some producers have started planting their field corn in various parts of the state. Planting is an important time to make decisions regarding herbicide selection for effective weed control throughout the field corn growing season. This article provides an overview and discuss some major herbicide options labelled in the NYS field corn.
Preplant burndown options
If no tillage is practiced, burndown herbicides such as glyphosate (Roundup PowerMax), glufosinate (Liberty), paraquat (Gramoxone), 2,4-D (2,4-D LV4) and saflufenacil (Sharpen) can be helpful to control winter annual weeds prior to corn planting. If glyphosate-resistant horseweed is present in the field, paraquat or combination of Sharpen + 2,4-D can be an effective burndown option. Make sure to use appropriate adjuvants as per each herbicide label to maximize the effectiveness of these burndown treatments. Burndown treatments should be made on actively growing winter annual weeds under optimum weather conditions (sunny conditions with air temperature above 55 F with no forecast of cold weather after applications).
Preemergence (PRE) herbicide options
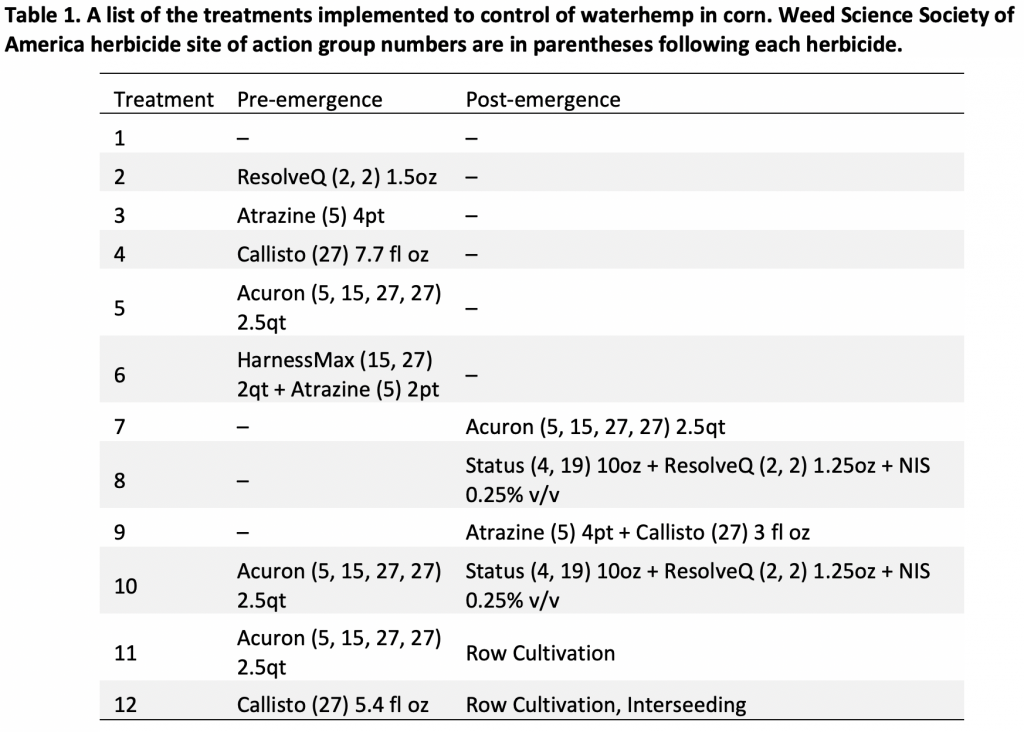
Preemergence or soil-applied herbicides are generally applied after crop planting but prior to its emergence. However, sometimes these preemergence herbicides can also be tank-mixed with preplant burndown treatments. Several preemergence options are available to use in field corn in the NY. Majority of these preemergence herbicides belong to Group 5, 14, 15, and 27 although there are few options from Group 2, 3, and 4 as well. Major preemergence herbicide options (not a complete list) along with their active ingredients and sites of action (SOA) labelled in NYS field corn are listed in Table 1. Several of these preemergence options are available in premixtures with two or three active ingredients from different groups (multiple SOA) and generally provide longer soil residual activity on summer annual weeds. For example, Harness Extra and FulTime NXT are premixtures of atrazine (Group 5) and acetochlor (Group 15) whereas Lumax EZ and Lexar EZ are premixtures of atrazine (Group 5), s-metolachlor (Group 15), and mesotrione (Group 27). Premixtures containing active ingredients from Group 5, 15 and 27 are most commonly used in field corn for grass and broadleaf weed control. While selecting appropriate preemergence option and its application rate, producers should thoroughly read the herbicide label for target weed species, rotational restrictions on the subsequent crops, cover crops or intercrops as well as consider the soil type, texture, and other soil properties (organic matter, soil pH, etc.).
Table 1. Preemergence herbicide options labelled in NY field corn.
| Herbicides | Active Ingredients | SOA |
| Prowl | Pendimethalin | 3 |
| Aatrex*¥ | Atrazine | 5 |
| Outlook*¥ | Dimethenamid | 15 |
| Surpass NXT*¥ | Acetochlor | 15 |
| Dual Magnum*¥ | S-metolachlor | 15 |
| Harness Xtra*¥, FulTime NXT*¥ | Atrazine + Acetochlor | 5, 15 |
| Bicep Lite II Magnum*¥, Cinch ATZ Lite*¥ | Atrazine + S-metolachlor | 5, 15 |
| Verdict*¥ | Saflufenacil + Dimethenamid | 14, 15 |
| Harness Max*¥ | Acetochlor + Mesotrione | 15, 27 |
| Acuron Flexi*¥ | S-metolachlor + Bicyclopyrone + Mesotrione | 15, 27 |
| Acuron*¥ | Atrazine + S-metolachlor + Bicyclopyrone + Mesotrione | 5, 15, 27 |
| SureStart II*¥ | Flumetsulam + Clopyralid + Acetochlor | 2, 4, 15 |
| Lumax EZ*¥, Lexar EZ*¥ | Atrazine + S-metolachlor + Mesotrione | 5, 15, 27 |
| Resicore*¥, Resicore XL*¥ | Clopyralid + Acetochlor + Mesotrione | 4, 15, 27 |
*Restricted Use Pesticides ¥Not for use in Nassau and Suffolk Counties
Postemergence (POST) herbicide options
Postemergence herbicides are applied after emergence of corn and weeds. Redroot pigweed, Powell amaranth, common lambsquarters, common ragweed, horseweed, common waterhemp, velvetleaf, foxtails (yellow, green, and giant), fall panicum, etc. are most common spring/summer annual weeds in NY corn. In addition, Palmer amaranth populations have also been recently found from six counties. In addition, field bindweeds, horsenettle, milkweed, yellow nutsedge, Canada thistle, hemp dogbane, quackgrass, etc. are most common perennial weeds. Johnsongrass populations have also been reported from corn fields in some southern counties of NY. Several postemergence herbicides are available to use in NY field corn to control these annual and perennial weed species. Majority of these labelled postemergence herbicides belong to Group 2, 4, 5, 6, 9, 10, 15, and 27.
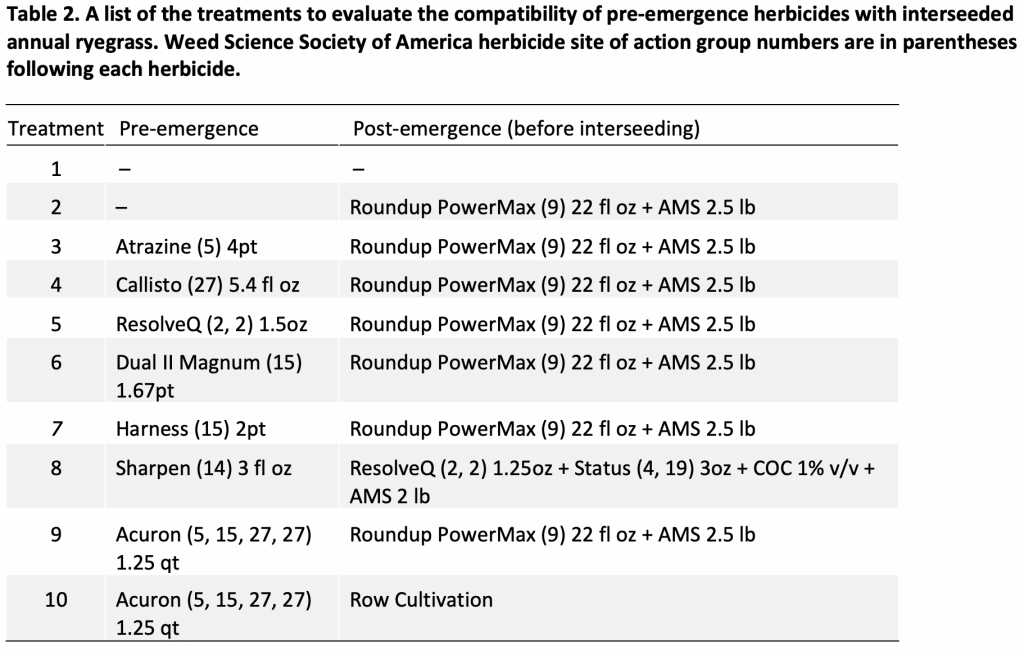
Table 2 highlights major postemergence herbicide options (not a complete list) along with their active ingredients and sites of action (SOA) labelled in conventional, Roundup Ready and Liberty Link corn hybrids. Several of these postemergence herbicides are broad-spectrum and can control both grass and broadleaf weed species. For instance, Postemergence applications of Capreno, Realm Q, Impact Core, Roundup PowerMax, Liberty can all help controlling grass and broadleaf weeds. In contrast, postemergence applied Aatrex, Banvel, Clarity, Callisto, Yukon are most effective controlling broadleaf weeds only. Producers should thoroughly read each herbicide label for target weed species, rotational restrictions on the subsequent crops, cover crops or intercrops during selection of appropriate postemergence option and its rate. Make sure to use appropriate adjuvants as per each herbicide label to maximize the effectiveness of these postemergence herbicides. If glyphosate- or triazine-resistant weeds are present, producers should select alternative effective two-pass herbicide program (preemergence followed by postemergence).
Table 2. Postemergence herbicide options labelled in NY field corn.
| Herbicides | Active Ingredients | SOA |
| For Conventional Corn Hybrids | ||
| Accent Q, Steadfast Q | Nicosulfuron, Nicosulfuron + Rimsulfuron | 2 |
| Permit, Resolve Q | Halosulfuron, Rimsulfuron + Thifensulfuron | 2 |
| Banvel, Clarity, DiFlexx | Dicamba | 4 |
| Aatrex*¥ | Atrazine | 5 |
| Basagran; Moxy 2EC | Bentazone; Bromoxynil | 6 |
| Callisto; Armezone/Impact; Laudis | Mesotrione; Topramezone; Tembotrione | 27 |
| Yukon | Halosulfuron + Dicamba | 2, 4 |
| Capreno*¥ | Thiencarbazon + Tembotrione | 2, 27 |
| Realm Q | Rimsulfuron + Mesotrione | 2, 27 |
| Impact Core*¥ | Acetochlor + Topramezone | 15, 27 |
| Kyro*¥ | Clopyralid + Acetochlor + Topramezone | 4, 15, 27 |
| For Roundup Ready Corn Hybrids | ||
| Roundup PowerMax; Durango DM | Glyphosate | 9 |
| Halex GT | Glyphosate + S-metolachlor+ Mesotrione | 9, 15, 27 |
| For Liberty Link Corn Hybrids | ||
| Liberty | Glufosinate | 10 |
*Restricted Use Pesticides ¥Not for use in Nassau and Suffolk Counties
Note: For further information on currently labelled PRE and POST herbicide options in NY field corn, check the 2024 Cornell Guide for Integrated Field Crop Management (available online).
Field Study in 2023
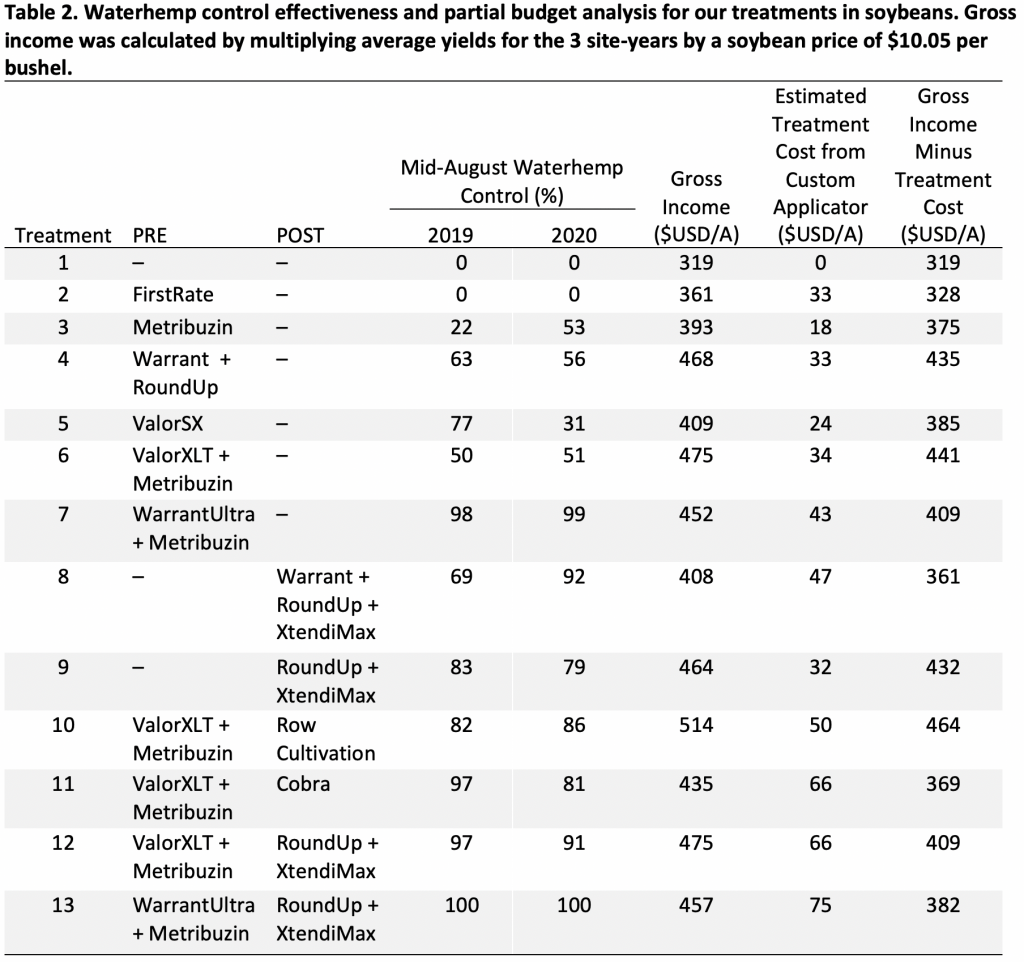
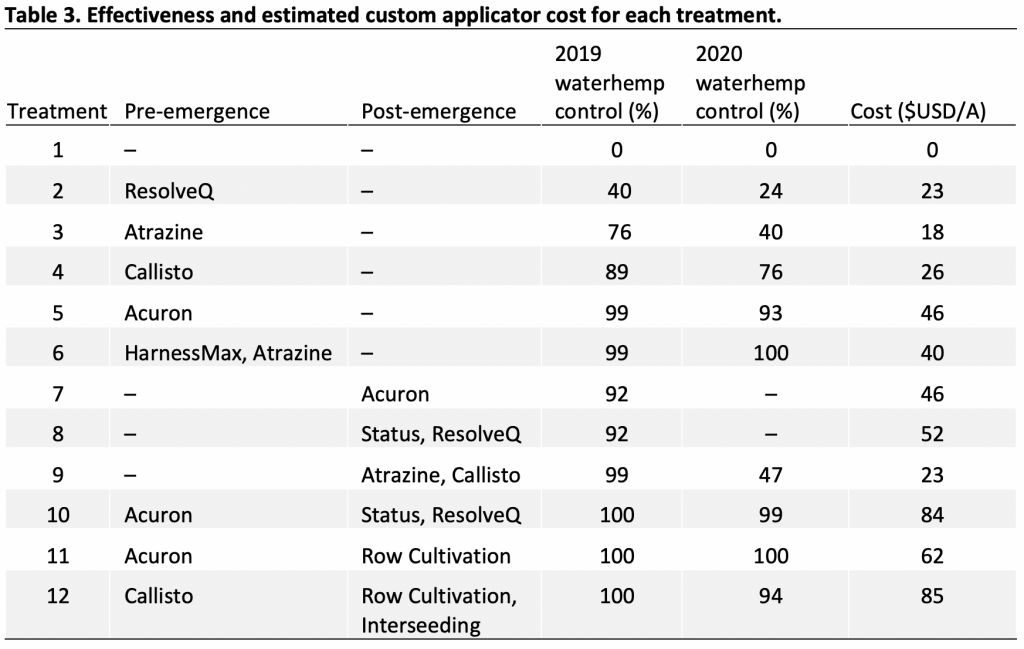
A field study was conducted in Franklin and Jefferson counties, NY, in 2023 growing season to determine the effectiveness of various preemergence herbicides (Table 3) with and without atrazine (42 fl oz/a) for weed control in field corn. Both field sites had natural infestation of common lambsquarters. Field corn was planted around May 20 at both sites and selected preemergence herbicides were applied immediately after planting. Small plots (10 feet wide by 30 feet long) were used to test each herbicide program. Test plots were laid arranged in Randomized Complete Block Design (RCBD) with 4 replications. All PRE herbicides were applied using CO2-operated backpack sprayer equipped with handheld boom with four nozzles (AIXR 110015). Results indicated no significant differences in common lambsquarters control among all tested preemergence herbicides at 35 days after treatments. Across both sites in Franklin and Jefferson counties, preemergence applied Acuron Flexi, Harness Max, Resicore XL, and Verdict + Outlook alone or with atrazine provided 94 to 100% control of common lambsquarters (Table 3; Figure 1)). In 2024, we plan to evaluate these preemergence applied herbicides (with or without atrazine) across multi-locations again to validate these results.
Table 3. Percent common lambsquarters control at 35 days after applications of various preemergence herbicide premixes alone or in combination with atrazine in field corn during 2023 growing season in Franklin and Jefferson Counties, NY.
| Herbicide | Active Ingredient (s) | Site of action (SOA) | Rate (oz/a) | Franklin | Jefferson |
| % control | |||||
| Acuron Flexi | S-metolachlor/bicyclopyrone/mesotrione | 15, 27 | 72 | 98 | 97 |
| Aatrex 4L + Acuron Flexi | Atrazine + S-metolachlor/bicyclopyrone/mesotrione | 5,15,27 | 42 + 72 | 99 | 94 |
| Harness Max | Acetochlor/mesotrione | 15,27 | 64 | 100 | 97 |
| Aatrex 4L + Harness Max | Atrazine + acetochlor/mesotrione | 5,15,27 | 42 + 64 | 99 | 97 |
| Resicore XL | Clopyralid/acetochlor/mesotrione | 4,15,27 | 96 | 99 | 98 |
| Aatrex 4L + Resicore XL | Atrazine + clopyralid/acetochlor/mesotrione | 5,4,15,27 | 42 + 96 | 100 | 99 |
| Verdict + Outlook | Saflufenacil/dimethenamid-P + dimethenamid-P | 14,15,15 | 16 + 4.6 | 99 | 94 |
| Aatrex 4L + Verdict + Outlook | Atrazine + saflufenacil/dimethenamid-P + dimethenamid-P | 5,14,15,15 | 16 + 4.6 + 42 | 99 | 98 |

Disclaimer: Brand names appearing in this publication are for product identification purposes only. Persons using such products assume responsibility for their use in accordance with current label directions of the manufacturer.