Biomicrofluidics, 2015: Enhancing sensitivity and specificity in rare cell capture microdevices with dielectrophoresis
Citation: Smith JP, Huang C, Kirby BJ, Enhancing sensitivity and specificity in rare cell capture microdevices with dielectrophoresis, Biomicrofluidics, 9:014116, 2015. doi pdf
Abstract: The capture and subsequent analysis of rare cells, such as circulating tumor cells from a peripheral blood sample, has the potential to advance our understanding and treatment of a wide range of diseases. There is a particular need for high purity (i.e., high specificity) techniques to isolate these cells, reducing the time and cost required for single-cell genetic analyses by decreasing the number of contaminating cells analyzed. Previous work has shown that antibody-based immunocapture can be combined with dielectrophoresis (DEP) to differentially isolate cancer cells from leukocytes in a characterization device. Here, we build on that work by developing numerical simulations that identify microfluidic obstacle array geometries where DEP-immunocapture can be used to maximize the capture of target rare cells, while minimizing the capture of contaminating cells. We consider geometries with electrodes offset from the array and parallel to the fluid flow, maximizing the magnitude of the resulting electric field at the obstacles’ leading and trailing edges, and minimizing it at the obstacles’ shoulders. This configuration attracts cells with a positive DEP (pDEP) response to the leading edge, where the shear stress is low and residence time is long, resulting in a high capture probability; although these cells are also repelled from the shoulder region, the high local fluid velocity at the shoulder minimizes the impact on the overall transport and capture. Likewise, cells undergoing negative DEP (nDEP) are repelled from regions of high capture probability and attracted to regions where capture is unlikely. These simulations predict that DEP can be used to reduce the probability of capturing contaminating peripheral blood mononuclear cells (using nDEP) from 0.16 to 0.01 while simultaneously increasing the capture of several pancreatic cancer cell lines from 0.03–0.10 to 0.14–0.55, laying the groundwork for the experimental study of hybrid DEP-immunocapture obstacle array microdevices.
Figures:
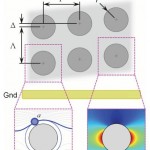
- FIG. 1. Antibody-functionalized obstacle arrays in a 2D microfluidic device can be used to engineer differential particle transport. This paper studies the effect of DEPassisted immunocapture, generated by applying an AC electric field to electrodes offset from the array and parallel to the direction of fluid flow. Target cells undergoing positive DEP (pDEP) are attracted to the high electric field magnitude regions at the obstacles leading and trailing edges (right inset), where the shear stress is low and the residence time is long (supporting the capture of the target cells); although they are also repelled from the low field magnitude regions at the obstacle shoulders, the locally high shear stress and short residence time minimizes the impact on overall capture. Likewise, contaminating cells undergoing negative DEP (nDEP) are repelled from regions where capture is likely (i.e., the obstacles’ leading edge) and attracted to regions where capture is unlikely (i.e., the obstacles’ shoulder).
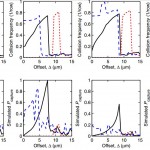
- FIG. 2. DEP alters cell trajectories within the microfluidic device, leading to changes in the mean collision frequency for cells within a given device geometry. Advection dominates DEP at the obstacles’ shoulder, but the reverse is true at the obstacles’ leading and trailing edges, where the fluid flow stagnates; as such, a cell’s response in the high electric field magnitude region at the leading and trailing edges has the most effect on its trajectory through the array. For medium and large cells (e.g., diameters B and C in this figure), pDEP attracts the cells to the high field magnitude regions near the leading and trailing edges, increasing the mean collision frequency and the time in contact (which supports capture), whereas nDEP (fCM < 0) repels cells from these regions. Likewise, pDEP forces small diameter cells (e.g., diameter A) toward the region of high field magnitude, increasing collision frequency compared to without DEP, but the overall collision frequency remains low. Although nDEP does indeed repel these small cells from the high field magnitude regions, nDEP displaces particle diameter A enough to cause a brief “grazing” cell-obstacle collision, increasing the collision frequency; these grazing events are brief and occur where the shear stress is highest, so capture of these cells is unlikely.
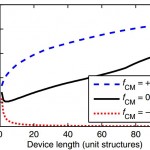
- FIG. 3. Modest displacements due to hFDEPi are amplified in the obstacle array geometry. This amplification increases with increasing device length, increasing the mean collision frequency (i.e., the collision frequency for all particles of a given diameter averaged over the length of the device) for particles with fCM > 0 and decreasing the collision frequency for particles with fCM < 0.
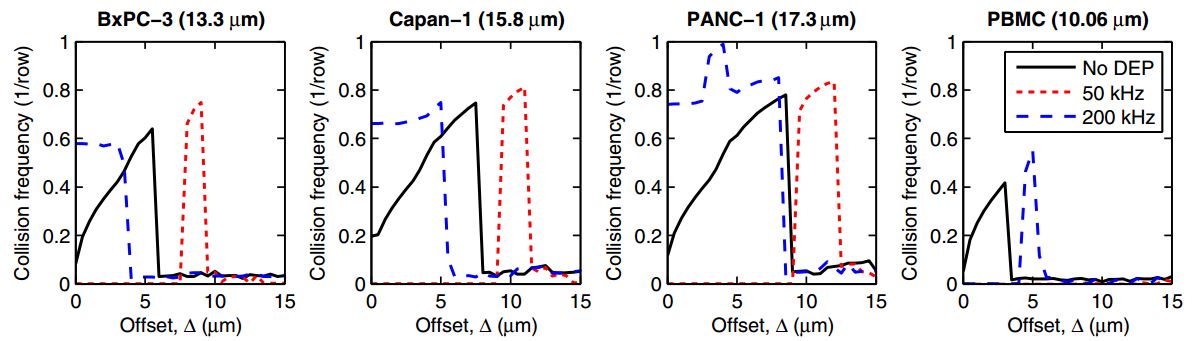
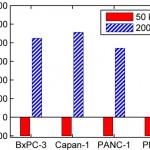
- FIG. 4. Simulated collision frequency (top row) and capture probability (Pcapture, bottom row) are shown vs. offset (D) for four cell types: BxPC-3, Capan-1, and PANC-1 pancreatic cancer cells; and PBMCs. At 50 kHz, all of the cells experience nDEP. At 200 kHz, the pancreatic cells experience pDEP and are attracted to the obstacles, increasing Pcapture; simultaneously, PBMCs experience nDEP at 200 kHz and are repelled from the obstacles, decreasing Pcapture. Note that Pcapture for PANC-1 is much lower than the other pancreatic cancer cells due its smaller A value (see Table I) and the displacement of some PANC-1 cells onto high collision frequency, high shear stress trajectories at 200 kHz.
- FIG. 5. DEP significantly effects the capture probability of cells in the obstacle array. Here, the percent change in simulated Pcapture is shown for pancreatic cancer cells and PBMCs for two independent simulations, one driven at 50 kHz and another at 200 kHz. The simulations show that the capture of contaminating PBMCs is reduced by greater than 99% at 200 kHz (from 0.16 to 0.001); simultaneously, the capture of target pancreatic cancer cells is increased by 370%–450% (from 0.03–0.10 to 0.14–0.55, based on cell line). Although the absolute capture probability for pancreatic cancer cells would be greater at a larger offset, the improvement in sample purity (i.e., the large reduction in Pcapture for PBMCs) predicted using DEP at a small offset outweigh a small decrease in capture efficiency, especially for applications such as single-cell genetic analyses.