Electrophoresis, 2008: Zeta potential and electroosmotic mobility in microfluidic devices fabricated from hydrophobic polymers: 1. The origins of charge
Citation: Tandon V, Bhagavatula SK, Nelson WC, Kirby BJ. Zeta potential and electroosmotic mobility in microfluidic devices fabricated from hydrophobic polymers: 1. The origins of charge, Electrophoresis, 29(5): pages 1092-1101. doi pdf
Abstract: This paper combines new experimental data for electrokinetic characterization of hydrophobic polymers with a detailed discussion of the putative origins of charge at water-hydrophobe interfaces. Complexities in determining the origin of charge are discussed in the context of design and modeling challenges for electrokinetic actuation in hydrophobic microfluidic devices with aqueous working fluids. Measurements of interfacial charge are complicated by slip and interfacial water structuring phenomena (see Part 2, this issue). Despite these complexities, it is shown that (i) several hydrophobic materials, such as Teflon and Zeonor, have predictable electrokinetic properties and (ii) electrokinetic data for hydrophobic microfluidic systems is most consistent with the postulate that hydroxyl ion adsorption is the origin of charge.
Figures:
- Figure 1. Scheme of the electrical double layer.
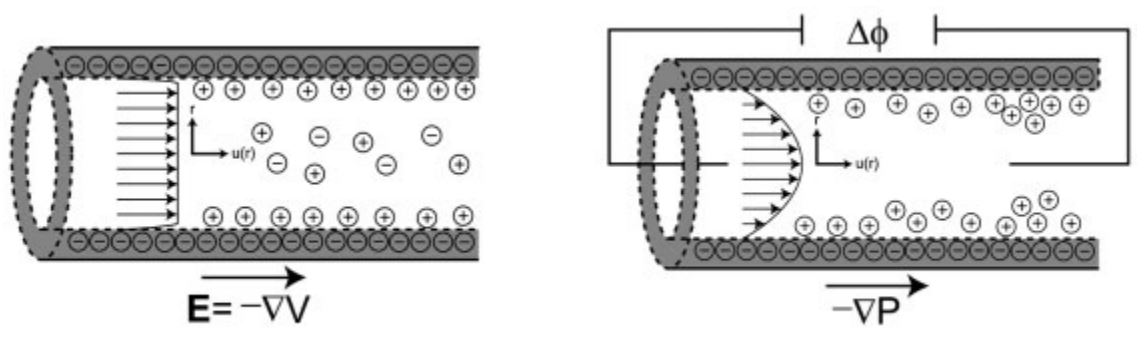
- Figure 2. (a) Scheme of uniform electroosmotic flow resulting from an applied electric field. (b) Scheme of the redistribution of ions in pressure driven flow in a microchannel, resulting in a streaming potential. Ion sizes and distributions in the schematics are not to scale.
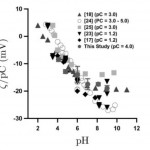
- Figure 3. z normalized by pC (= – log C, C is the counterion concentration in M) as a function of pH for PTFE. Modified from [6] with addition of recent current monitoring data from the authors.
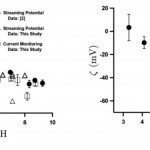
- Figure 4. (a) z normalized by pC= – log C (C is the counterion concentration in M) as a function of pH for Zeonor. (b) Streaming potential data showning z as a function of pH for TOPAS (pC = 3)