Optics Express, 2008: Microfluidic devices for terahertz spectroscopy of biomolecules
Citation: Paul A. George, Wallace Hui, Farhan Rana, Benjamin G. Hawkins, A. Ezekiel Smith, Brian J. Kirby. Microfluidic devices for terahertz spectroscopy of biomolecules, Optics Express, 16(3): pages 1577-1582. doi pdf
Abstract: We demonstrate microfluidic devices for terahertz spectroscopy of biomolecules in aqueous solutions. The devices are fabricated out of a plastic material that is both mechanically rigid and optically transparent with near-zero dispersion in the terahertz frequency range. Using a lowpower terahertz time-domain spectrometer, we experimentally measure the absorption spectra of the vibrational modes of bovine serum albumin from 0.5 – 2.5 THz and find good agreement with previously reported data obtained using large-volume solutions and a high-power free-electron laser. Our results demonstrate the feasibility of performing high sensitivity terahertz spectroscopy of biomolecules in aqueous solutions with detectable molecular quantities as small as 10 picomoles using microfluidic devices.
Figures:
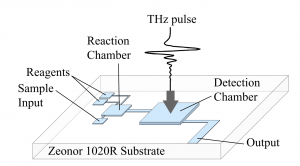
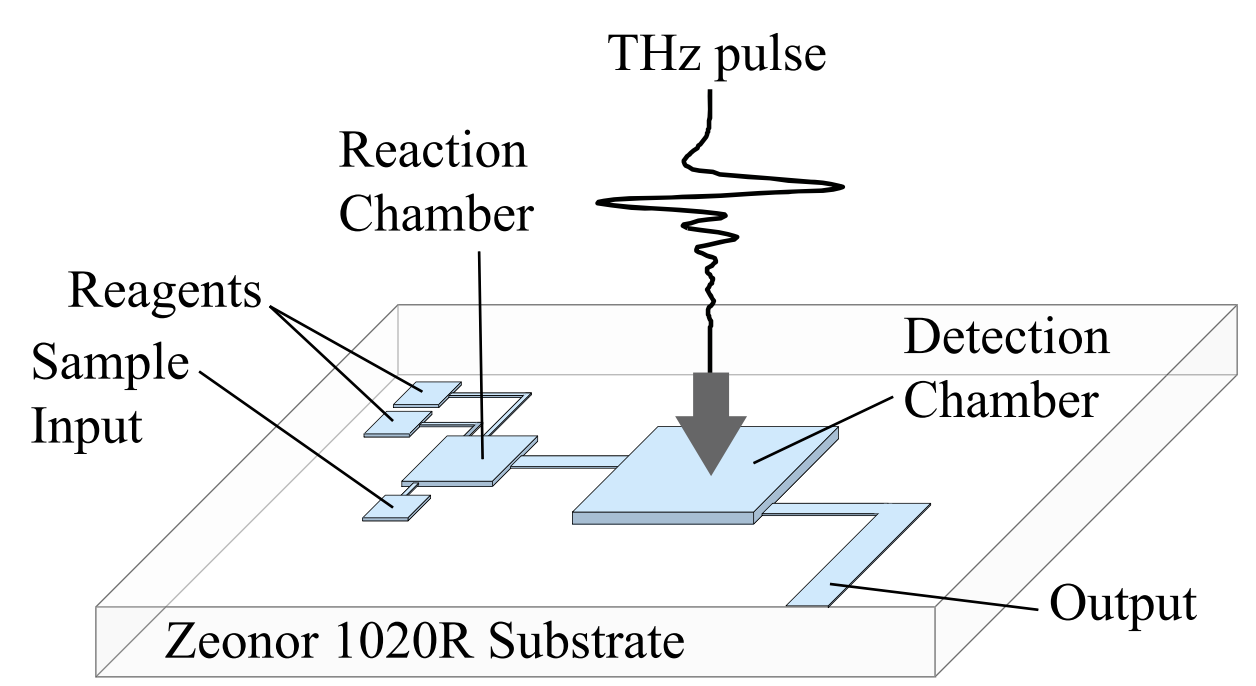
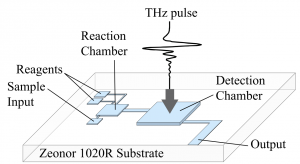
Fig. 1. A system for on-chip THz sensing. The device contains sample and reagent inputs, a
reaction chamber, and a detection chamber for THz as well as optical and IR spectroscopy
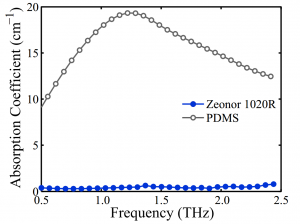
Fig. 2. The measured absorption coefficient of Zeonor 1020R and PDMS. Zeonor has a
nearly constant index of 1.518 (not shown) and an absorption coefficient <1 cm−1 at THz
frequencies, which is 10-20 times smaller than that of PDMS.
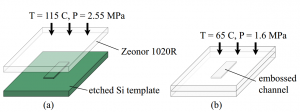
Fig. 3. Fabrication of the microfluidic devices used in this work. (a) A slab of Zeonor 1020R
was first embossed using a Si template. (b) The embossed slab was then bonded to another
piece of Zeonor 1020R. The final channel depth was 95 micro-meters.
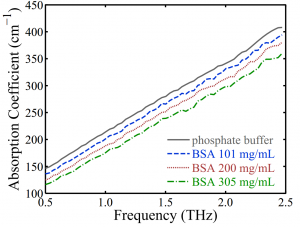
Fig. 4. The absorption coefficient of the phosphate buffer and BSA solutions measured by
THz-TDS using microfluidic devices. Values are extracted using Eq. 2
![Fig. 5. (a) The measured molecular absorption coefficient of hydrated BSA molecules. In agreement with Beer’s Law, the absorption coefficient does not depend on solution concentration. (b) The molar extinction of BSA measured using microfluidic channels compared to the results in [7]. The excellent agreement demonstrates the feasibility of performing THz spectroscopy of biomolecules in microfluidic channels using low-power THz sources.](https://blogs.cornell.edu/kirbyresearch/files/2016/10/2008_George_Opticsexpress_5-1k34vb0-300x118.png)
Fig. 5. (a) The measured molecular absorption coefficient of hydrated BSA molecules. In
agreement with Beer’s Law, the absorption coefficient does not depend on solution concentration.
(b) The molar extinction of BSA measured using microfluidic channels compared
to the results in [7]. The excellent agreement demonstrates the feasibility of performing
THz spectroscopy of biomolecules in microfluidic channels using low-power THz sources.
- Fig. 1. A system for on-chip THz sensing. The device contains sample and reagent inputs, a reaction chamber, and a detection chamber for THz as well as optical and IR spectroscopy
- Fig. 2. The measured absorption coefficient of Zeonor 1020R and PDMS. Zeonor has a nearly constant index of 1.518 (not shown) and an absorption coefficient <1 cm−1 at THz frequencies, which is 10-20 times smaller than that of PDMS.
- Fig. 3. Fabrication of the microfluidic devices used in this work. (a) A slab of Zeonor 1020R was first embossed using a Si template. (b) The embossed slab was then bonded to another piece of Zeonor 1020R. The final channel depth was 95 micro-meters.
- Fig. 4. The absorption coefficient of the phosphate buffer and BSA solutions measured by THz-TDS using microfluidic devices. Values are extracted using Eq. 2
- Fig. 5. (a) The measured molecular absorption coefficient of hydrated BSA molecules. In agreement with Beer’s Law, the absorption coefficient does not depend on solution concentration. (b) The molar extinction of BSA measured using microfluidic channels compared to the results in [7]. The excellent agreement demonstrates the feasibility of performing THz spectroscopy of biomolecules in microfluidic channels using low-power THz sources.