Modulation of BMP signaling
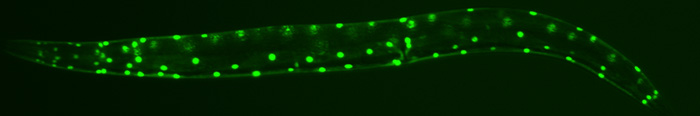
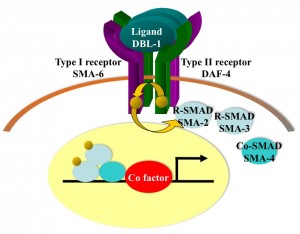
Bone morphogenetic proteins (BMPs) belong to the transforming growth factor beta (TGFbeta) superfamily of ligands and the BMP signaling pathway plays roles in multiple developmental and homeostatic processes. Malfunction of the pathway causes many somatic and hereditary disorders in humans, including cardiovascular diseases and cancer. We are interested in uncovering mechanisms involved in modulating and ensuring proper spatiotemporal control of BMP signaling.
Our lab has devised a unique and sensitive genetic screen to identify new components of the BMP pathway. Using this screen, we have identified multiple evolutionarily conserved factors that function in modulating BMP signaling, which includes DRAG-1/RGM, UNC-40/neogenin/DCC, SUP-17/ADAM-10, SMOC-1, and three tetraspanins (TSP-21, -12 and -14). Except for the secreted SMOC-1 protein, all of the other BMP modulators are integral membrane or membrane-associated proteins that appear to function in the receptor-expressing cells. We are investigating mechanistically how each of these factors functions in regulating BMP signaling, and testing whether their functions are conserved in mammals. We are also actively engaged in identifying new factors modulating BMP signaling.
Mesoderm patterning and fate diversification
The mesoderm germ layer gives rise to a diverse array of functionally important cell types, including various types of muscle and non-muscle cells. While the early stages of mesoderm determination are increasingly well understood, the underlying basis of later myogenic and non-myogenic diversity within the mesoderm remains largely unknown. We are interested in dissecting the regulatory networks underlying the diversification and cell fate specification of the mesoderm.
We use the C. elegans postembryonic mesoderm lineage, the M lineage, as a model system for these studies. The M lineage is derived from a single pluripotent precursor cell, the M mesoblast. In a hermaphrodite animal during postembryonic development, the M mesoblast divides in a characteristic and reproducible pattern to generate 14 striated bodywall muscles required for locomotion, 16 non-striated sex muscles (of four different types) used for egg-laying, and 2 non-muscle macrophage-like coelomocytes involved in heavy metal detoxification. The M lineage is therefore a unique system for studies of how different mesodermal fates are diversified from a single progenitor cell, how lineage information becomes correlated with myogenic determination, and how diverse programs of asymmetric (dorsoventral and anterioposterior) patterning, cell division timing and orientation, and cell fate specification are integrated.
We have a collection of mutants with defects in M lineage development. Studying the corresponding genes for some of these have allowed us to gain insight into the regulatory networks underlying M lineage development. Most of the factors that we have identified are conserved in vertebrates, suggesting similar mechanisms shared by C. elegans and vertebrates in mesoderm diversification. Current efforts are geared towards identifying the genetic and epigenetic mechanisms involved in M lineage diversification.